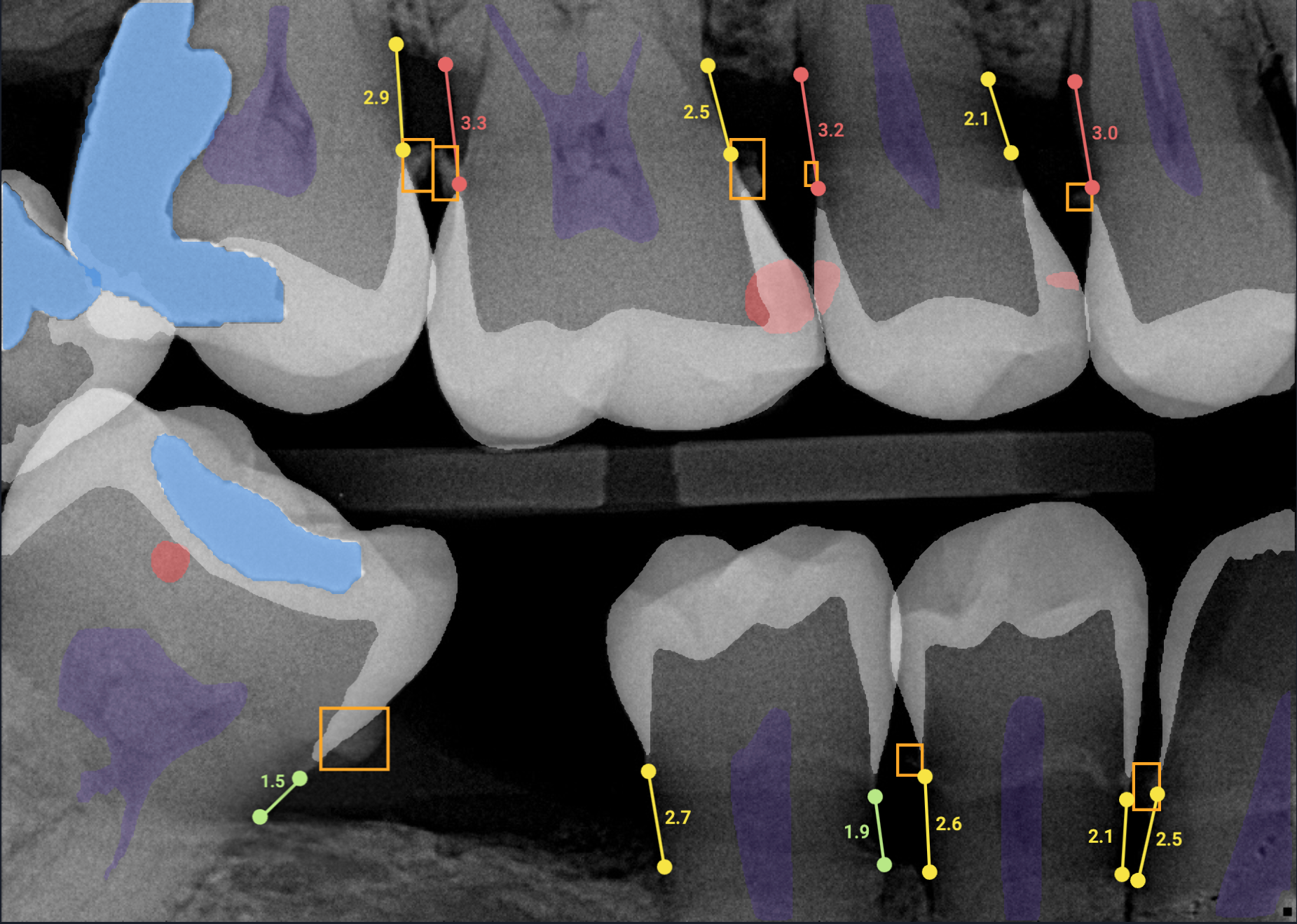
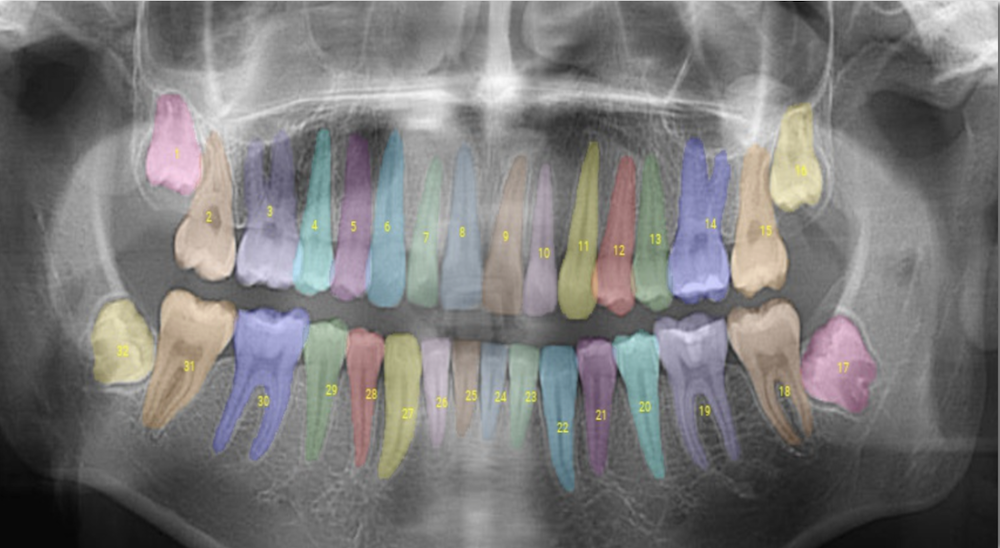
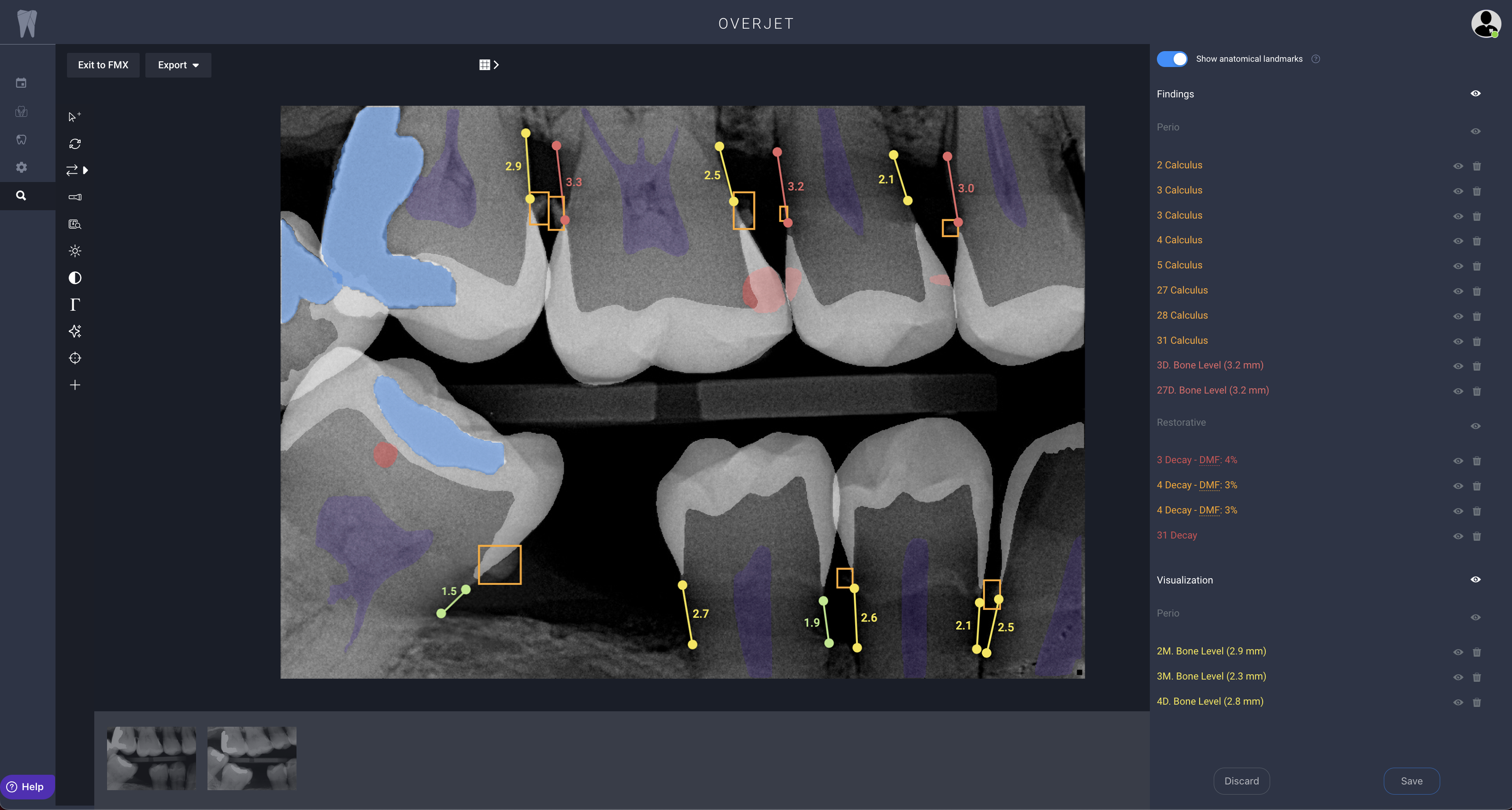
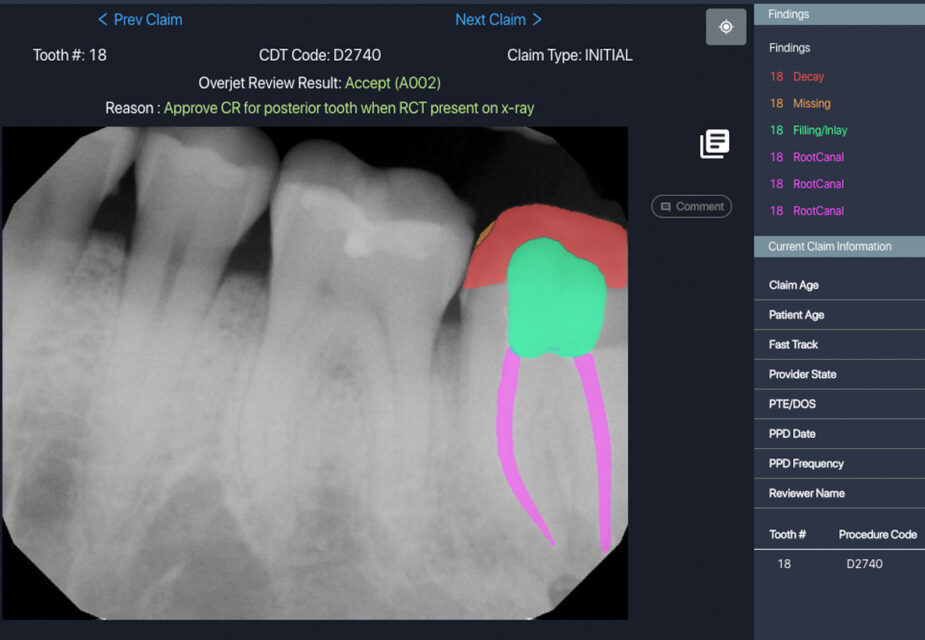
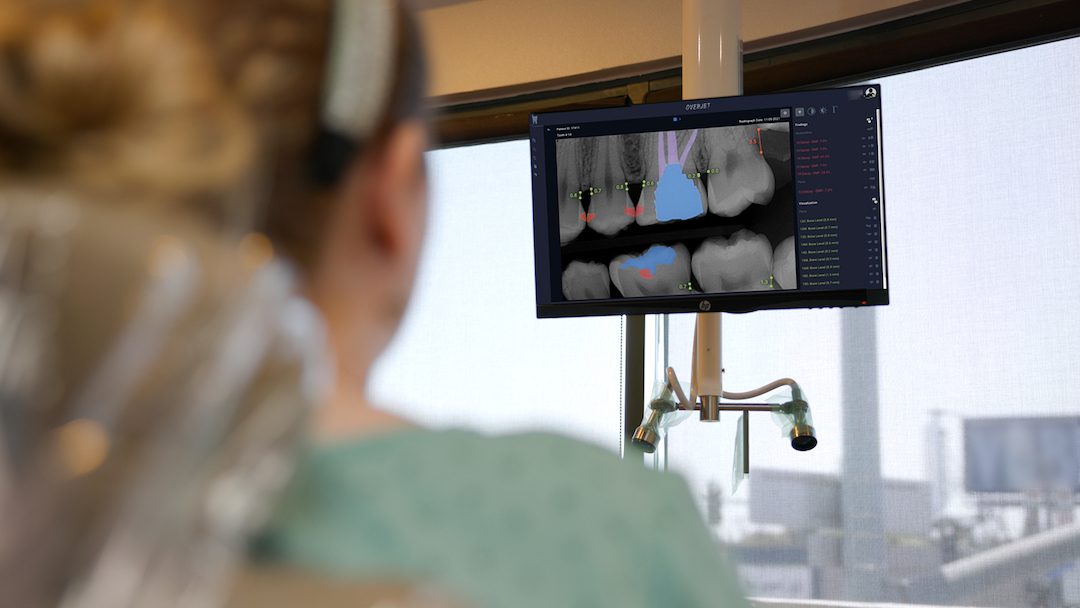
Overjet evaluates clinical performance by combining clinical AI findings in radiographs with patient and treatment data from the practice management system. These findings are then used by clinical staff to improve clinical precision, efficiency, training, and communication, allowing practices to achieve consistent and better patient outcomes.
Perfect for: dental groups, group practices, DSOs